



Nicholas Schott, Graduate Student (Stegemann and Putnam Laboratories), Biomedical Engineering, University of Michigan
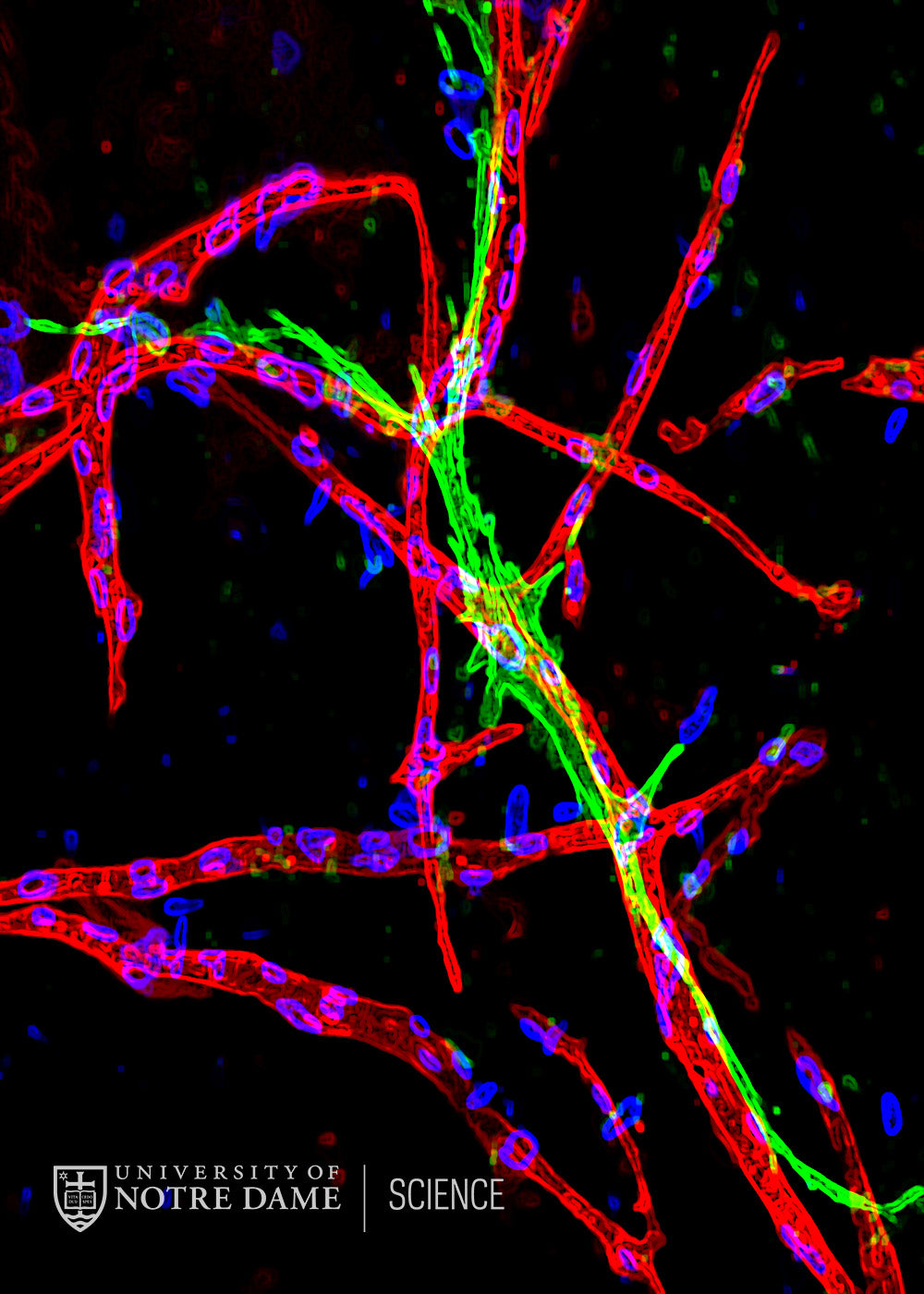
Organoids and engineered tissues made in the laboratory hold major therapeutic promise, but a bottleneck to their successful use is that they rarely contain blood vessels (vasculature). Pre-vascularizing such constructs represents a promising means to circumvent this shortcoming, but achieving vasculature that is functionally mature is a critical challenge. This image depicts an innovative strategy: coculture of endothelial cells (which make blood vessels) with mesenchymal stem cells (MSC, which can give rise to muscle). Both cell types are embedded within a fibrin hydrogel and cultured in the lab for several weeks. Over this time course, endothelial cells assemble into highly branched, multicellular structures (red). MSC then support subsequent maturation of the newly vascular network by forming direct interdigitations with endothelial cells and regulating vessel permeability. This stimulus in turn induces MSC to differentiate into smooth muscle (green), which reinforces the vessel walls.
20-091
